ONWARD Medical N.V. (Euronext: ONWD), the medical technology company creating innovative therapies to restore movement, independence, and health in people with spinal cord injury (SCI), today announces the first patient enrollment in the HemON Study and first-in-human use of the Company’s ARCIM implantable pulse generator (IPG), designed to stimulate the spinal cord to restore movement and autonomic function for people with spinal cord injury and other conditions that affect mobility.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20220509005325/en/
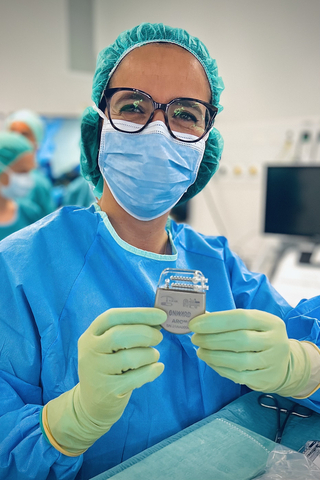
Neurosurgeon Dr. Jocelyne Bloch holding ARC-IM IPG. Credit: ONWARD Medical
“ONWARD’s ARCIM IPG offers surgeons precision and flexibility never before available in an implantable neurostimulator,” said Dr. Jocelyne Bloch, Chief of Functional Neurosurgery at Switzerland’s Lausanne University Hospital (CHUV). “Clinicians will be able to leverage these advanced capabilities to refine and deliver epidural stimulation therapies for people with spinal cord injury.”
The ONWARD ARCIM IPG was purpose-designed to deliver targeted electrical stimulation to the spinal cord in the precise areas responsible for triggering or controlling movement and autonomic functions that may be affected by a spinal cord injury or neurodegenerative disorder. The IPG is designed to operate in closed-loop, incorporating data from sensors or other devices that may be deployed inside or outside the body. It is also designed to deliver therapy through an associated ARCIM lead with precision and flexibility, allowing clinicians to apply biomimetic stimulation that can closely replicate normal spinal cord activity patterns during mobility or autonomic function.
The HemON Study (NCT05111093) aims to evaluate the safety and preliminary efficacy of ARCIM Therapy to improve blood pressure management and trunk control in people with spinal cord injury who suffer from orthostatic hypotension, which is characterized by debilitatingly low blood pressure that may occur when people sit upright, stand, or change body position. Orthostatic hypotension has been observed in approximately 75% of people with spinal cord injury. HemON will enroll up to 16 participants at CHUV in Lausanne, Switzerland.
“The first implant of our ARCIM IPG is a huge milestone for ONWARD and the SCI community,” said Dave Marver, CEO of ONWARD. “This important achievement validates our vision for a future in which clinicians will no longer need to struggle with modified pain stimulators to explore new treatments and instead will be able to use devices and therapies designed specifically to treat people with spinal cord injury and their unique needs.”
To learn more about ONWARD’s ARC Therapy and the company’s vision to restore movement, independence and health in people with spinal cord injury, please visit ONWD.com.
THIS PRESS RELEASE CONTAINS INSIDE INFORMATION WITHIN THE MEANING OF ARTICLE 7(1) OF THE EUROPEAN MARKET ABUSE REGULATION (596/2014).
About ONWARD
ONWARD is a medical technology company creating innovative therapies to restore movement, independence, and health in people with spinal cord injury. ONWARD’s work builds on more than a decade of basic science and preclinical research conducted at the world’s leading neuroscience laboratories. ONWARD’s ARC Therapy, which can be delivered by implantable (ARCIM) or external (ARCEX) systems, is designed to deliver targeted, programmed stimulation of the spinal cord to restore movement and other functions in people with spinal cord injury, ultimately improving their quality of life. ONWARD has received three Breakthrough Device Designations from the FDA encompassing both ARCIM and ARCEX. The company’s first FDA pivotal trial, called Up-LIFT, completed enrollment in December 2021 with 65 subjects worldwide.
ONWARD is headquartered at the High Tech Campus in Eindhoven, the Netherlands. It maintains an office in Lausanne, Switzerland and has a growing U.S. presence in Boston, Massachusetts, USA. For additional information about the company, please visit ONWD.com. To access our 2022 Financial Calendar, please visit IR.ONWD.com.
Disclaimer
Certain statements, beliefs and opinions in this press release are forward-looking, which reflect the Company or, as appropriate, the Company directors’ current expectations and projections about future events. By their nature, forward-looking statements involve a number of risks, uncertainties and assumptions that could cause actual results or events to differ materially from those expressed or implied by the forward-looking statements. These risks, uncertainties and assumptions could adversely affect the outcome and financial effects of the plans and events described herein. A multitude of factors including, but not limited to, changes in demand, competition and technology, can cause actual events, performance or results to differ significantly from any anticipated development. Forward looking statements contained in this press release regarding past trends or activities should not be taken as a representation that such trends or activities will continue in the future. As a result, the Company expressly disclaims any obligation or undertaking to release any update or revisions to any forward-looking statements in this press release as a result of any change in expectations or any change in events, conditions, assumptions or circumstances on which these forward-looking statements are based. Neither the Company nor its advisers or representatives nor any of its subsidiary undertakings or any such person’s officers or employees guarantees that the assumptions underlying such forward-looking statements are free from errors nor does either accept any responsibility for the future accuracy of the forward-looking statements contained in this press release or the actual occurrence of the forecasted developments. You should not place undue reliance on forward-looking statements, which speak only as of the date of this press release.
View source version on businesswire.com: https://www.businesswire.com/news/home/20220509005325/en/
For Media Enquiries:
media@onwd.com
Simon Gentry +44 (0)20 3757 6772
For Investor Enquiries:
investors@onwd.com
For Company Enquiries:
ONWARD – info@onwd.com
Permalink: http://www.businesswire.com/news/home/20220509005325/en
COMUNICATO STAMPA - Responsabilità editoriale di Business Wire







